The only report with 99% physician agreement1-3
Curated reports are generated by our CE-marked and FDA cleared advanced AI, Zio™ ECG Utilization Service (ZEUS) system, and verified by Qualified ECG Specialists with 99% physician agreement.1,4-7

Reporting
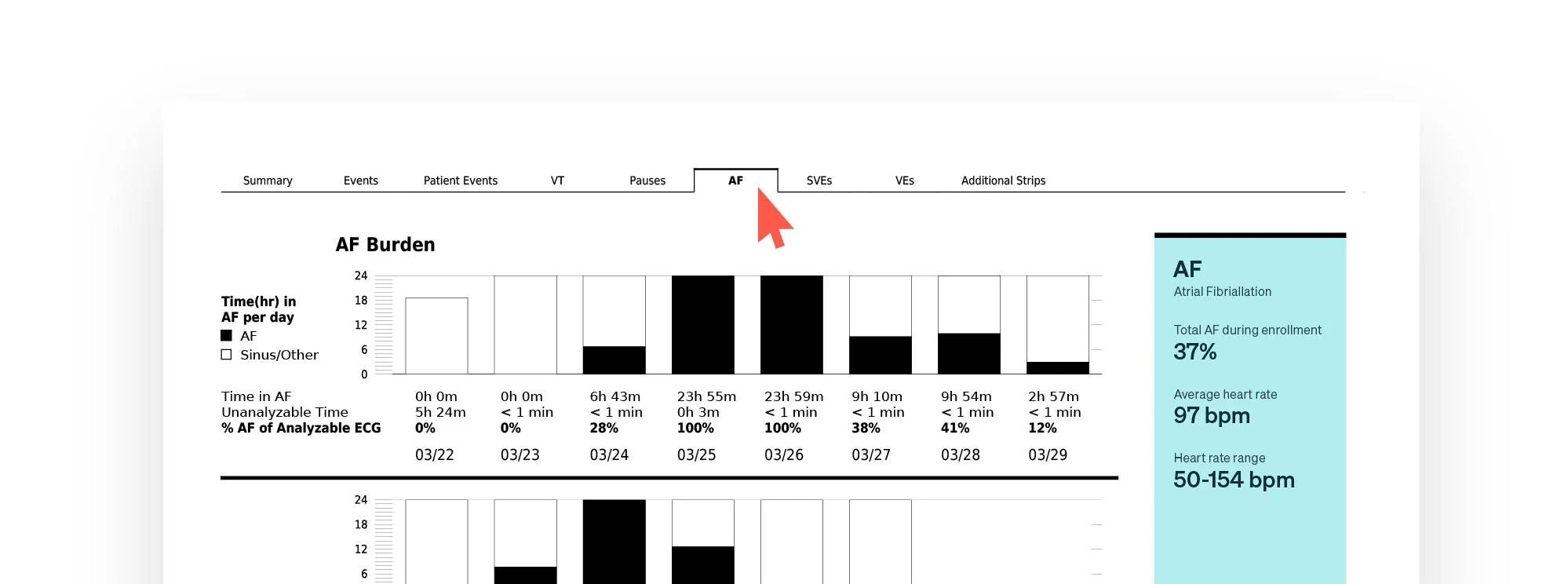
1. The Zio™ Report cover page provides a snapshot of actionable and clinically significant findings. The upper-right hand corner of the report details the wear time and the total analyzable time with artifacts removed.
2. Sample strips are arranged in order of clinical significance with additional at-a-glance information such as duration, episodes, and burdens.
3. Navigate with one click from the cover page to any of the report sections for more details. Download the report for the full experience.
4. The Zio Preliminary Findings are generated by a CE-marked and FDA-cleared deep-learned algorithm* and each report is validated by Qualified ECG Specialists (QES).4-7
Get a comprehensive Zio Report

Advanced deep-learned algorithm*
The Zio report is generated by a CE-marked and FDA-cleared deep learned algorithm that can detect 13+ different types of arrhythmias, plus sinus rhythm and artifacts, just as well as expert, board-certified cardiologists.4-7
10 million patient records8-9
2 billion hours of curated heartbeat data10-11
CE-marked and FDA-cleared deep learned algorithm4-7
Detects 13+ types of arrhythmias4-7

The human side of the equation.
Because of our commitment to accuracy, our team of Qualified ECG Specialists reviews and curates 100% of our reports before you receive them.
As elegant and easy to navigate as it is powerful.
The Zio report provides you with a rich summary of the most important cardiac events right on the first page — and an easy-to-navigate deep dive for all the intricate details captured.
- 99% of physicians agree with the comprehensive end-of-wear report. Based on a review of all online Zio XT, Zio monitor, and Zio AT end-of-wear reports. Data on file. iRhythm Technologies, 2023.
- Based on the US data.
- Zio service provides continuous, uninterrupted recording and a comprehensive end-of-wear report.
- Data on file. iRhythm Technologies, 2020.
- Hannun et al. Cardiologist-level arrhythmia detection and classification in ambulatory electrocardiograms using a deep neural network. Nat Med. 2019;25:65-69. https://doi.org/10.1038/s41591-018-0268-3
- Deep learned algorithm is only available in the United States, European Union, Switzerland, United Kingdom, and Japan.
- FDA 510K clearance, CE mark, UKCA mark, and PMDA-approval.
- Data on file. iRhythm Technologies. 2025.
- Based on the US data using Zio ECG monitors.
- Data on file. iRhythm Technologies, 2024.
- Based on the US and UK data using Zio® ECG monitors.
*ZioTM ECG Utilization Service (ZEUS) System
The Zio™ service is a long-term continuous ambulatory cardiac monitoring service. It consists of the Zio™ monitor that collects beat-to-beat ECG data and can be worn for up to 14 days; the Zio™ ECG Utilization Service (ZEUS) System deep learned algorithm, which detects arrhythmias. The data is reviewed and curated by a team of Qualified ECG Specialists (QES) to provide the final patient report.
For indications for use, warnings & contraindications, visit here.
iRhythm, Zio, Zio monitor, and ZioSuite are trademarks of iRhythm Technologies, Inc. ©2024 All rights reserved.
Zio™ monitor and ZEUS are CE-marked (CE2797).
Zio monitor, ZEUS and Zio service are currently available in Austria, Netherlands, Spain & Switzerland.
For further regulatory information visit here.
WEB0209.01